12.1 Karst Landscapes and Systems
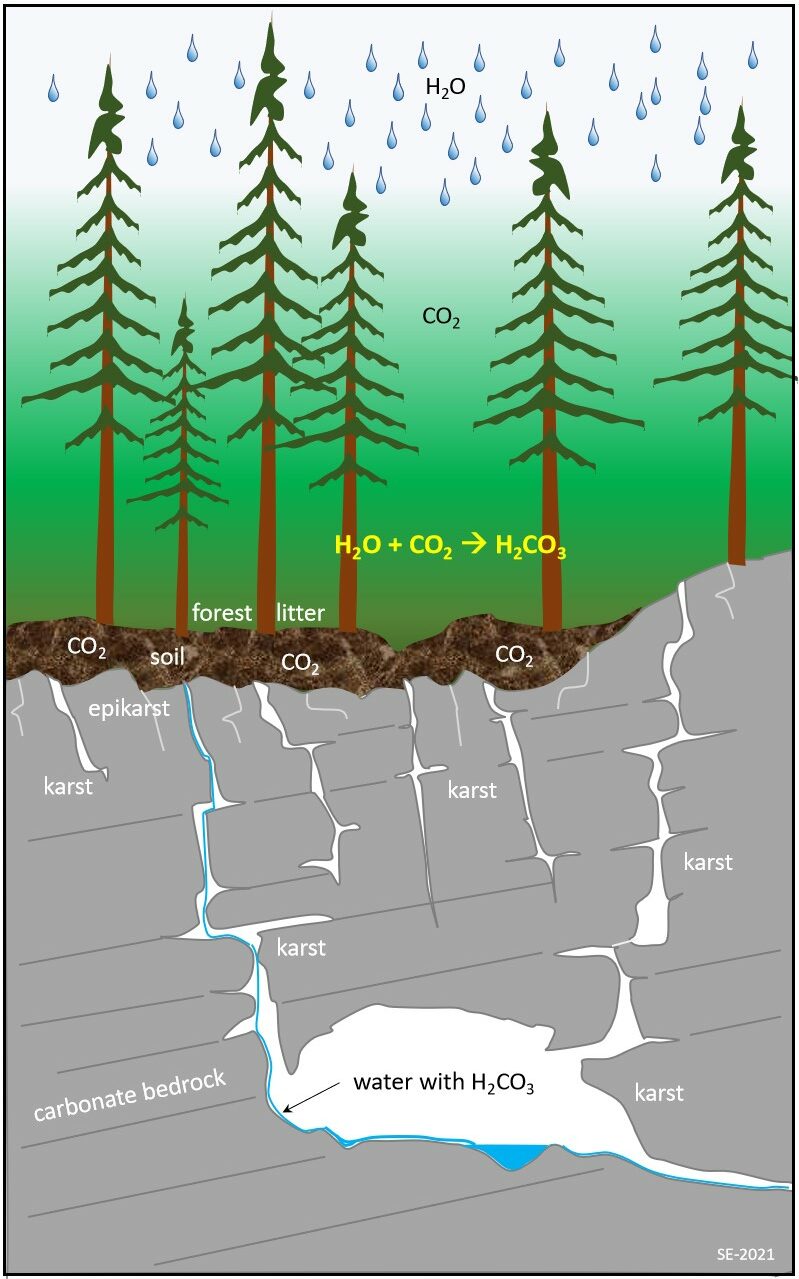
Karstification is a process dominated by chemical dissolution of soluble bedrock (Figure 12.1.1). It starts as carbon dioxide from the atmosphere dissolves in rainwater falling to the surface of the Earth. The water becomes further enriched in carbon dioxide as it infiltrates the soil, and the result is slightly acidic surface water and groundwater. Slightly acidic water when in contact with limestone (or other soluble bedrock types) promotes a chemical reaction which slowly dissolves the bedrock. Existing fractures or crevices in the rock are preferentially widened forming larger cracks allowing for more water flow and dissolution. As the cracks widen, mechanical erosion takes place as loose rock fragments transported by water rub against the sides of the openings, some of which eventually form caves.
A critical requirement for the development of karst is water. Without water there would be no karst or caves! Carbon-dioxide (CO2) is another key component as it dissolves in water forming a weak carbonic acid solution (H2CO3) as shown below. This carbonic acid reacts with the solid limestone (predominately CaCO3) to form the ions Ca2+ and HCO3.
H2O + CO2 -> H2CO3
CaCO3 + H2CO3 -> Ca2+ + 2HCO3–
Several other factors also play important roles in the development of karst such as: the type and nature of the soluble bedrock, the thickness and type of soil cover, and the hydraulic head or difference in elevation from top to bottom of a karst landscape (Figure 12.1.2). Some of the prime bedrock attributes that play a role in karst development include chemical purity, fracturing, thickness, and geometrical shape. In general, the greater the percentage of calcite (CaCO3) in a limestone, the greater the potential for dissolution. Fracturing in karst bedrock enhances the flow of water and provides preferential sites for conduit development. The thickness of the soluble bedrock unit as well as its geometrical configuration (e.g., tilted, folded, interbedded) can determine the three-dimensional shapes of the karst landscapes.

Soils can play a significant role in the karst processes. Soils that are rich in organic matter (e.g., a forest floor or a swamp) have higher levels of CO2 and so make rainfall or surface waters more acidic, thus enhancing the dissolution process. Thick relatively impermeable soils such as compacted glacial till drift cover can protect soluble bedrock types from dissolution. Likewise, glacial tills rich in carbonate material can buffer rainfall or surface water reducing its natural acidity.
Hydraulic head, or the elevation change in the topography, drives water flow. A soluble bedrock unit that extends all the way from a high elevation to a low elevation (i.e., has a great topographic relief) is likely to have a greater potential for karst development than one that occurs at a uniform lower elevation.
Karst Values, Systems, and Ecosystems
Karst is important for a wide variety of reasons. On a global scale a significant portion (15-20%) of the Earth’s surface is underlain by limestone (and other soluble bedrock types) that have the potential to form karst.[1] An understanding of karst processes is therefore important, particularly where humans interact with this landscape. Karst landscapes have certain features and resource values that are not present in non-karst landscapes. Karst aquifers provide the main source of water in many parts of the world, for example, 25% of US groundwater come from karst.[2]
Karst caves are sites for unique subterranean habitats, many of which have yet to be explored and studied. Caves are also depositories (or storage sites) for critical information on past life forms (fossils), ancient cultures and paleoclimates.
As in many issues related to Earth science and other life sciences, it is important to learn and understand about karst using a systems approach – where the karst system is a three-dimensional landscape comprised of:
- interlinked sub-components of the karst geosphere (bedrock and soil)
- karst hydrosphere
- karst atmosphere
- karst biosphere
There is a continual interchange of materials and energy between these sub-components (Figure 12.1.3). Without this approach we are not able to fully understand the processes of karstification nor appreciate the values of this resource. Karst landscapes can also include their own and unique ecosystems. An ecosystem is where complex group of organisms (plants, animals, fungi, and micro-organisms) live together with their surrounding environment of rock, soil, air, water, and nutrients.
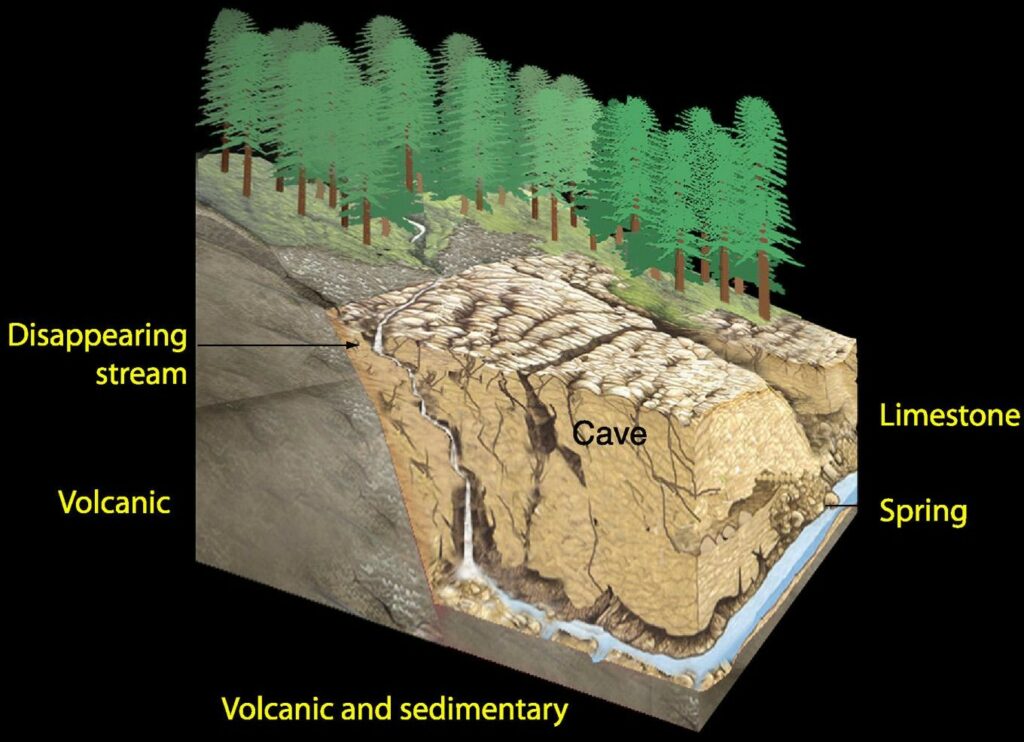
Karst ecosystems or karst biota refers to all the plants and animals living in, or using, karst. This includes the flora and fauna of surface epikarst cavities, cave ecosystems, cave entrance zones or large sinkholes, as well as the broader karst landscape. Some components of the karst biota, such as some cave dwellers, are highly specialized that they are unable to live outside their underground niches. Others maybe opportunistic and colonize karst environments or utilize karst features. Bats, for example, can use certain caves for roosts or sites of hibernation. Other surface-dwelling animals, such as deer and bears, may use sinkholes and cave entrance zones for thermoregulation as such sites tend to be cooler in the heat of summer or warmer in winter.
Karst aquatic systems can be highly productive, and fish may use cave and karst conduit systems for spawning, shade, or as protection from predators. Stable and cool water temperatures can be regulated by underground residence times in karst systems, providing suitable environment for a range of aquatic organisms.
Karst Around the World
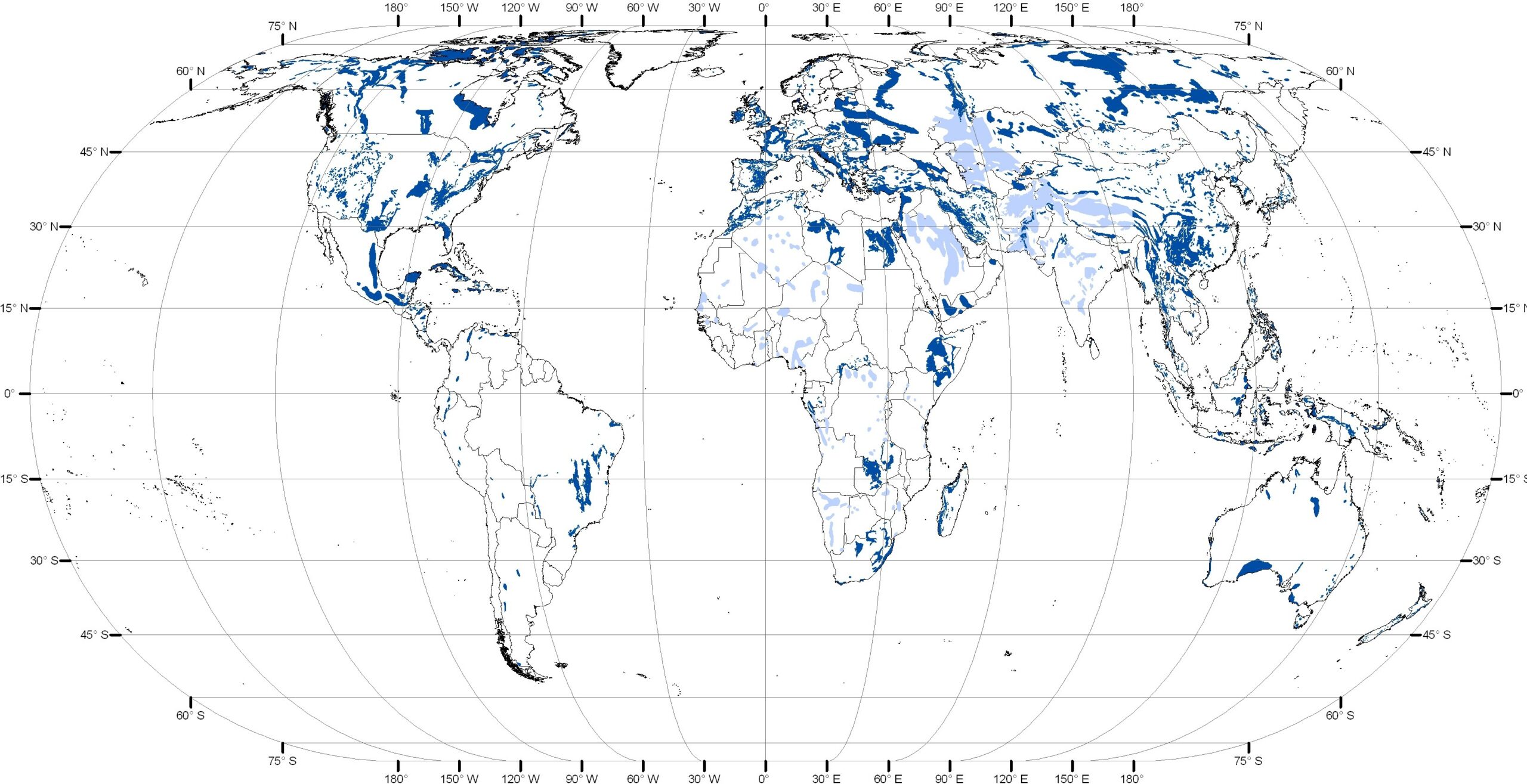
Karst occurs throughout the world (Figure 12.1.4). Approximately 20% of the land area underlain by carbonate bedrock has the potential to form karst, [3] although almost a third of that is considered unsuitable for present day karst development due to unfavorable climate, burial with overlying materials, and low relief. Some of the most ‘karst-rich’ regions of the world include Southeast Asia, Europe, Central America, Southeast US.
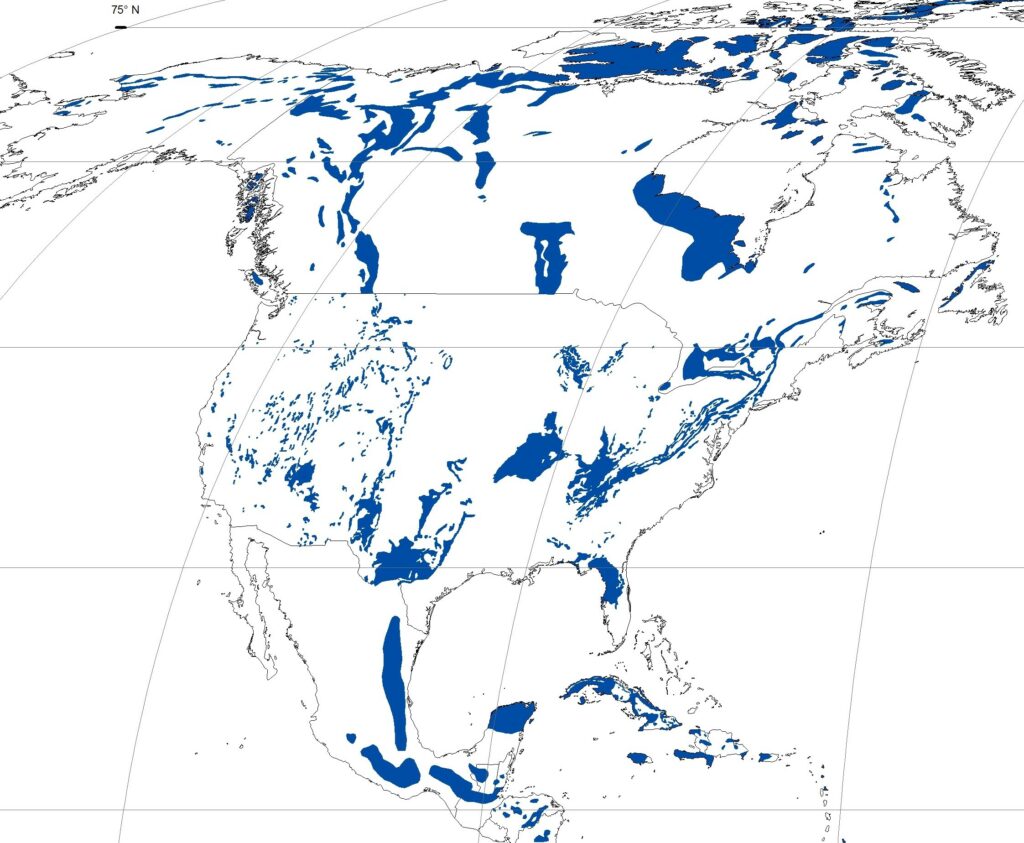
In North America karst of some form or another can be found in almost all the provinces, territories and states (Figure 12.1.5). Canada has one of the widest ranges of karst types in the world. Approximately 10-15% of Canada is underlain by limestone, dolomite and evaporate rocks (halite and gypsum). [4] Examples of karst types include gypsum karst in Nova Scotia, halite (or rock salt) karst in Saskatchewan, forested karst in coastal British Columbia, alpine karst in the Canadian Rockies, and arctic karst in the far north.
Approximately 10% of British Columbia is underlain by carbonate rocks with the potential for karst.[5] Some of the best-known karst occurs along the Pacific Coast—primarily Vancouver Island and Haida Gwaii and occurs within temperate rainforest. This forested karst is characterized by abundant rainfall, mild temperatures, well developed surface karst features, cave systems, large coniferous trees, thick understory vegetation, mature soils of glacial and organic origin (Figures 12.1.6).

Media Attributions
- Figure 12.1.1 Steven Earle, CC BY 4.0, after the BC Ministry of Forests, 1997.
- Figure 12.1.2 Soil and Fractured Bedrock by T. Stokes, CC BY 4.0 (Stokes et al., 2011)
- Figure 12.1.3 3D Karst Model by T. Stokes, CC BY 4.0 (Stokes et al., 2011)
- Figure 12.1.4 Williams, P., Fong Y. T. (n.d.). Karst: World Map of Carbonate Rock Outcrops v3.0. School of Environment, University of Auckland. https://www.fos.auckland.ac.nz/our_research/karst/)
- Figure 12.1.5 (Williams & Fong, n.d.)
- Figure 12.1.6 Photo by T. Stokes, CC BY 4.0
- Ford, D., Williams, P. (2007). Karst hydrology and geomorphology. John Wiley & Sons. ↵
- Weary, D. & Doctor, D. ()2012). Karst in the United States: A digital map compilation and database. US Geological Survey (USGS) Open-File Report 2012-1156. https://pubs.usgs.gov/of/2014/1156/pdf/of2014-1156.pdf ↵
- (Ford & Williams, 2007). ↵
- Ford, D. (2004). Canada. In Gunn, J. (Ed.), Encyclopedia of caves and karst science (p. 359). Fitzroy Dearborn. ↵
- Stokes, T. R., & Griffiths, P. A. (2019). An overview of the karst areas in British Columbia, Canada. Geoscience Canada, 46(1), 49–66. https://doi.org/10.12789/geocanj.2019.46.145 ↵
